Age-related macular degeneration (AMD) is a leading cause of vision loss, affecting millions worldwide. The disease, which primarily impacts older adults, progresses in two forms: dry AMD, which gradually damages the macula, and wet AMD, a more severe condition involving abnormal blood vessel growth. Recent advances in research and treatment are offering new hope for patients, with developments in nutritional support, gene therapy, stem cell research, and long-acting drug delivery systems reshaping AMD management.
Key Takeaways
A clinical trial is investigating stem cell therapy for advanced dry age-related macular degeneration (AMD), offering new hope for patients who currently have no FDA-approved treatment options.
- Researchers at the National Eye Institute (NEI) are conducting a Phase I/IIa trial to assess the safety and feasibility of transplanting retinal pigment epithelial (RPE) cells derived from a patient’s own induced pluripotent stem cells.
- The trial involves implanting an iPSC-derived RPE patch beneath the retina to help slow the progression of vision loss associated with geographic atrophy.
- Eligible participants must be 55 years or older, have AMD-related geographic atrophy in at least one eye, and meet specific visual acuity criteria.
Stem cell transplantation for GA
Geographic atrophy, the advanced stage of dry age-related macular degeneration (AMD), leads to progressive vision loss with no FDA-approved treatment currently available. Researchers at the National Eye Institute (NEI) are now exploring stem cell therapy as a potential solution. This clinical trial aims to determine whether transplanting retinal pigment epithelium (RPE) cells derived from a patient’s own stem cells can help slow disease progression.
A groundbreaking Phase I/IIa clinical trial
This pioneering trial aims to evaluate the safety and feasibility of transplanting RPE cells generated from a patient’s own induced pluripotent stem cells (iPSCs). The process involves taking adult cells, such as blood cells, and reprogramming them into stem cells. These stem cells are then developed into RPE cells, which are critical for maintaining photoreceptors, the retina’s light-sensing cells.
In the trial, an iPSC-derived RPE patch measuring approximately 2×4 mm is implanted under the retina of the worse-seeing eye. The patch is grown on a biodegradable scaffold, allowing it to integrate with existing retinal structures. Researchers aim to determine whether this approach can help slow the progression of vision loss associated with geographic atrophy.
The study will initially enroll five participants, each with a best-corrected visual acuity (BCVA) between 20/100 and 20/500 in the affected eye. If no major safety concerns arise, the trial will expand to include up to seven additional participants with a BCVA between 20/80 and 20/500. Patients will be closely monitored over five years to evaluate both safety and potential benefits.
Addressing the challenges of dry AMD treatment
Unlike wet AMD, which involves abnormal blood vessel growth, dry AMD leads to the gradual degeneration of RPE and photoreceptor cells. The loss of these cells results in the formation of geographic atrophy, creating blind spots in central vision.
Traditional efforts to treat dry AMD have faced significant hurdles. RPE transplants must integrate successfully under the retina without triggering an immune response. Additionally, ensuring the long-term survival and functionality of transplanted cells has proven difficult. By using a patient’s own stem cells, this trial seeks to minimize rejection risks and explore the potential for personalized medicine in retinal disease treatment.
Clinical trial participation
Eligibility requirements for participants: The trial is recruiting up to 12 participants aged 55+ with AMD-related geographic atrophy in at least one eye.
Eligible participants must have a BCVA of 20/100 to 20/500 (first cohort) or 20/80 to 20/500 (second cohort) in the affected eye. They cannot have wet AMD, prior treatment, glaucoma, or high intraocular pressure. Good overall health is required for surgery. RPE transplantation will be performed only in the worse-seeing eye. While those with severe vision loss may not see improvement, the study aims to slow disease progression and preserve vision.
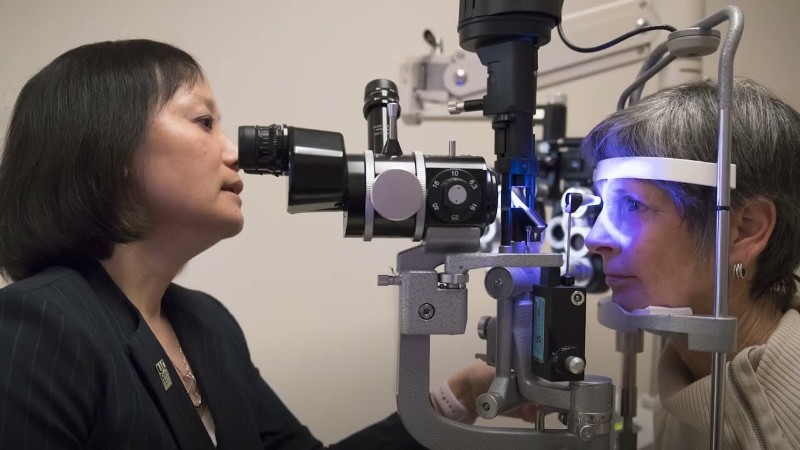
Screening and evaluation process: Before enrollment, potential participants must undergo a comprehensive screening process at the NEI Eye Clinic. This evaluation includes a full eye exam, including visual acuity testing, retinal imaging, and fluorescein angiography, a medical assessment, including blood tests, urinalysis, a chest X-ray, an EKG, and other necessary evaluations, and a review of prior medical history and any other eye-related conditions.
Those who pass the screening will be scheduled for surgery, with the RPE patch taking approximately six months to manufacture.
Breakthroughs in Dry AMD treatment
Until recently, there were no FDA-approved treatments for geographic atrophy (GA), the advanced stage of dry AMD. However, recent approvals and ongoing trials are changing the landscape of GA treatment.
FDA-approved therapies for Geographic Atrophy
Syfovre (Apellis Pharmaceuticals): The first FDA-approved treatment for GA, Syfovre works by targeting the C3 complement pathway, a part of the immune system that contributes to retinal cell degeneration. Clinical trials demonstrated that Syfovre slows lesion growth and could help preserve vision for longer.
Izervay (Astellas/Iveric Bio): Another FDA-approved GA therapy, Izervay blocks the C5 complement pathway, reducing lesion expansion and slowing disease progression.
Stem cell therapy and retinal repair
Stem cell research has shown promising early results in restoring vision for AMD patients by replacing damaged retinal cells.
The London Project at Moorfields Eye Hospital successfully transplanted retinal pigment epithelial (RPE) cells derived from embryonic stem cells into AMD patients. Some individuals saw vision improvements lasting over a year.
Regenerative Patch Technologies has developed a stem-cell-derived RPE patch, which showed vision stabilization and improvement in early trials.
Luxa Biotechnology recently began testing RPE stem cell transplantation in a legally blind patient with dry AMD. The therapy involves implanting 50,000 RPE stem cells under the macula to support retinal regeneration.
As scientists continue to uncover the mechanisms behind AMD, emerging treatments could dramatically improve the outlook for patients. Gene therapy, stem cell transplantation, and extended-release drug delivery systems represent the future of AMD care, offering potential long-term solutions with fewer invasive procedures. With ongoing clinical trials and regulatory approvals, patients now have more options than ever to slow vision loss and maintain independence.